The incidence of high blood pressure among U.S. adults would be astounding if the correlations with poor lifestyle choices weren’t so clear. Obesity, poor nutrition, and inadequate exercise are all too common among adults, and they relate directly with unhealthy blood pressure levels according to the CDC. The latest CDC statistics claim about 1 in 3 adults have high blood pressure, and another 1 in 3 have prehypertension, meaning blood pressure higher than normal but not in the official high blood pressure range. Wearable tech developers are working to find ways to accurately measure blood pressure without using the traditional pressure cuff. We’ve seen a wide variety of contact and non-contact measurement methods. We’ve written about smartphone cases that purport to make cuff-less blood pressure readings and a wearable blood pressure monitor.
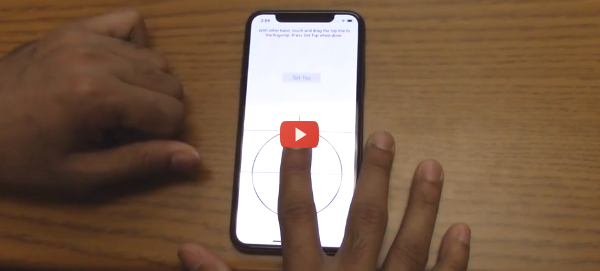
Michigan State University (MSU) researchers are working on an iPhone X app that can read blood pressure via oscillometric finger presses with no external hardware required. The same team, led by electrical and computer engineering professor Ramakrishna Mukkamala, previously developed the app-driven oscillometric method with external optical and force sensors. Newer smartphones with advanced capabilities allow the same functionality without additional hardware, Mukkamala says. The key is the iPhone X’s “peek and pop” 3D-touch gesture. In typical use, peek and pop allows you to see a bit of content (the peek). Then, if you want to see more, you can open an email by pressing on the screen to open the full message (the pop). This feature is made possible by a strain gauge built into the phone’s display so that it can detect varying degrees of pressure.
The MSU app guides the user to place and press a fingertip on the screen. The app reads the pressure with the phone’s strain gauge at the same time that it measures the blood volume oscillations using the phone’s camera. The MSU team tested the smartphone app with 20 subjects and published the results of the study in Scientific Reports. In 18 of the test cases, the blood pressure readings were close to finger cuff readings. Readings for two subjects were discarded because of finger placement issues, which would be a continuing concern.
The app needs to be clinically validated. Mukkamala said that because no extra hardware is required, once regulatory testing is complete the software should be able to proceed quickly through the approval stage and then be ready for consumer use. If indeed the MSU app works through the steps to approval, smartphones that support force detection could be employed worldwide to help consumers track their blood pressure quickly and easily.


COST OF SUCH A MOBILE WHICH MEASURES THE BP OF PATIENTS
If you search Health Tech Insider for “blood pressure,” you will find that we have covered a lot of newer technologies that measure blood pressure without a traditional pressure cuff. I expect that Valencell’s “in the ear” technology may be the first to come to market with a commercial product through one of their licensees, but there are other technologies that under development.
All the best,
Alfred Poor
Editor, Health Tech Insider